Chemistry, 08.04.2020 05:01 markitakimbrough69
A chemist prepares a solution by adding 293 mgmg of K2Cr2O7K2Cr2O7 (MWMW = 294.19 g/molg/mol ) to a volumetric flask, and then adding water until the total volume of the contents of the flask reaches the calibration line that indicates 250 mLmL . Determine the molarity of the prepared solution.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, Lesquirrel
Acar tire has a pressure of 2.38 atm at 15.2 c. if the pressure inside reached 4.08 atm, the tire will explode. how hot would the tire have to get for this to happen? report the temperature in degrees celsius.
Answers: 2

Chemistry, 22.06.2019 01:30, adrian08022
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 23.06.2019 00:00, familyk0jj3
How do you determine the percent yield of a chemical reaction
Answers: 1
You know the right answer?
A chemist prepares a solution by adding 293 mgmg of K2Cr2O7K2Cr2O7 (MWMW = 294.19 g/molg/mol ) to a...
Questions in other subjects:

Mathematics, 05.10.2020 01:01





Geography, 05.10.2020 01:01

Mathematics, 05.10.2020 01:01

History, 05.10.2020 01:01


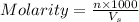
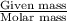
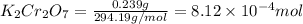
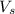 = volume of solution in ml = 250 ml
= volume of solution in ml = 250 ml