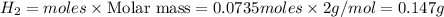Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
Sodium hydroxide reacts with aluminum and water to produce hydrogen gas:2 Al(s) + 2 NaOH(aq) + 6 H2O...
Questions in other subjects:



Biology, 29.08.2019 07:30


Social Studies, 29.08.2019 07:30



Computers and Technology, 29.08.2019 07:30

Business, 29.08.2019 07:30

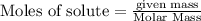
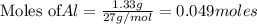
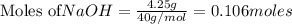
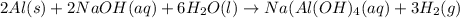
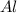 require = 2 moles of
require = 2 moles of ![NaOH/tex]Thus 0.049 moles of [tex]Al](/tpl/images/0589/0248/32375.png) will require=
will require=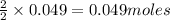 of
of 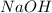
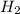
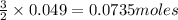 of
of