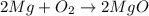Chemistry, 08.04.2020 04:45 AlysonDvz5464
Suppose two chemical reactions are linked together in a way that the O2 produced in the first reaction goes on to react completely with Mg to form MgO in the second reaction. Reaction one: 2 KClO3 → 3 O2 + 2 KCl Reaction two: 2 Mg + O2 → 2 MgO If you start with 4 moles of KClO3, how many moles of MgO could eventually form

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:50, kelonmazon2492
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

You know the right answer?
Suppose two chemical reactions are linked together in a way that the O2 produced in the first reacti...
Questions in other subjects:

Mathematics, 04.08.2019 02:30

History, 04.08.2019 02:30

Biology, 04.08.2019 02:30

Social Studies, 04.08.2019 02:30

Chemistry, 04.08.2019 02:30


History, 04.08.2019 02:30

Social Studies, 04.08.2019 02:30

Social Studies, 04.08.2019 02:30

 produced from 4 mole of
produced from 4 mole of 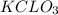

 mole of
mole of