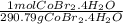Chemistry, 08.04.2020 02:56 sairaanwar67
Calculate the molarity of a solution obtained dissolving 10.0 g of cobalt(Ⅱ) bromide tetrahydrate in enough water to make 450 mL of solution
a. 6.80 × 10-2
b. 7.64 × 10-2
c. 7.64 × 10-5
d. 7.51 × 10-2
e. 0.102 M

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, miamassimino
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3
You know the right answer?
Calculate the molarity of a solution obtained dissolving 10.0 g of cobalt(Ⅱ) bromide tetrahydrate in...
Questions in other subjects:







Mathematics, 04.04.2020 11:42