
Chemistry, 08.04.2020 00:56 klmklm3799
Magnesium oxide can be made by heating magnesium metal in the presence of oxygen. The balanced equation for the reaction is: 2Mg(s)+O2(g)→2MgO(s) When 10.1 g of Mg are allowed to react with 10.5 g of O2, 10.3 g of MgO are collected. Dteremine the limiting reactants for the reaction

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

You know the right answer?
Magnesium oxide can be made by heating magnesium metal in the presence of oxygen. The balanced equat...
Questions in other subjects:



English, 14.07.2019 12:00





Biology, 14.07.2019 12:00


History, 14.07.2019 12:00

 = 10.1 g
= 10.1 g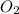 = 10.5 g
= 10.5 g = 32 g/mol
= 32 g/mol
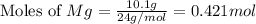



 moles of
moles of 

