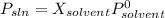Chemistry, 07.04.2020 23:47 mariamsakayanthebest
At a certain temperature, the vapor pressure of pure benzene is atm. A solution was prepared by dissolving g of a nondissociating, nonvolatile solute in g of benzene at that temperature. The vapor pressure of the solution was found to be atm. Assuming that the solution behaves ideally, determine the molar mass of the solute.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:40, markipler01
What is the study of how matter and energy interact? a. biology b. physics c. planetary science d. chemistry
Answers: 1

Chemistry, 22.06.2019 07:20, mathman783
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1


Chemistry, 23.06.2019 01:20, hflores0001
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
At a certain temperature, the vapor pressure of pure benzene is atm. A solution was prepared by di...
Questions in other subjects:

History, 09.06.2020 02:57

Business, 09.06.2020 02:57

Mathematics, 09.06.2020 02:57



History, 09.06.2020 02:57


Mathematics, 09.06.2020 02:57

Mathematics, 09.06.2020 02:57