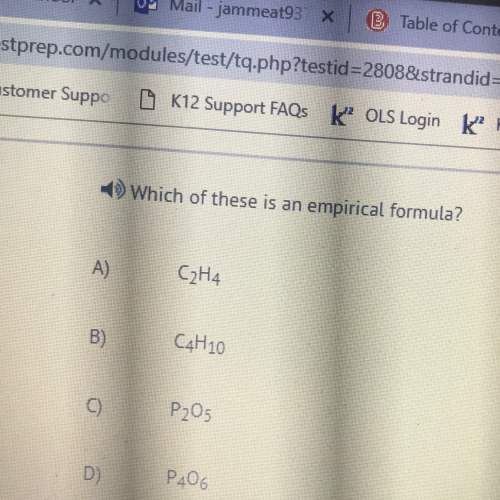
Chemistry, 07.04.2020 23:28 barry14201
20. Select the false statement below. A) Generally speaking, as you go across a period in the periodic table, the effective nuclear charge (Zeff) increases because each successive element contains more protons in the nucleus than the previous element and the outer electrons are all in the same shell as you go across a period. B) Generally speaking, the heavier halogens can be predicted to exhibit greater shielding of the outer-shell electrons by the inner-shell electrons than lighter halogens. C) Generally speaking, non-metallic elements have less effective nuclear charge (Zeff) than metallic elements in the same period. D) Generally speaking, as you go down a group in the periodic table, there is successively greater shielding of the outer-shell electrons by the inner-shell electrons.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 08:00, ggdvj9gggsc
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1


You know the right answer?
20. Select the false statement below. A) Generally speaking, as you go across a period in the period...
Questions in other subjects:

Mathematics, 17.02.2021 21:20

Spanish, 17.02.2021 21:20


Mathematics, 17.02.2021 21:20


History, 17.02.2021 21:20



English, 17.02.2021 21:20