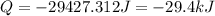Chemistry, 07.04.2020 23:35 maxy7347go
How much energy must be removed from a 94.4 g sample of benzene (molar mass= 78.11 g/mol) at 322.0 K to solidify the sample and lower the temperature to 205.0 K? The following physical data may be useful. ΔHvap = 33.9 kJ/mol ΔHfus = 9.8 kJ/mol Cliq = 1.73 J/g°C Cgas = 1.06 J/g°C Csol = 1.51 J/g°C Tmelting = 279.0 K Tboiling = 353.0 K

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:30, kiaramccurty
What type of organic molecule comprises the majority of a potato?
Answers: 1

You know the right answer?
How much energy must be removed from a 94.4 g sample of benzene (molar mass= 78.11 g/mol) at 322.0 K...
Questions in other subjects:


Mathematics, 26.08.2019 23:30

Mathematics, 26.08.2019 23:30




Arts, 26.08.2019 23:30

Computers and Technology, 26.08.2019 23:30

Chemistry, 26.08.2019 23:30

Mathematics, 26.08.2019 23:30

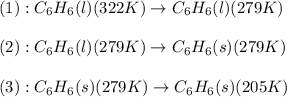
![Q=[m\times c_{p,l}\times (T_{final}-T_{initial})]+[m\times \Delta H_{fusion}]+[m\times c_{p,s}\times (T_{final}-T_{initial})]](/tpl/images/0587/9526/727a2.png)
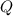 = heat released for the reaction = ?
= heat released for the reaction = ?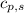 = specific heat of solid benzene =
= specific heat of solid benzene = 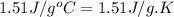
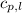 = specific heat of liquid benzene =
= specific heat of liquid benzene = 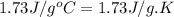
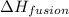 = enthalpy change for fusion =
= enthalpy change for fusion = 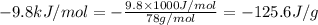
![Q=[94.4g\times 1.73J/g.K\times (279-322)K]+[94.4g\times -125.6J/g]+[94.4g\times 1.51J/g.K\times (205-279)K]](/tpl/images/0587/9526/0dc0d.png)