Chemistry, 08.04.2020 00:09 chrismed2001
Lead(II) nitrate is added slowly to a solution that is 0.0800 M in Cl− ions. Calculate the concentration of Pb2+ ions (in mol / L) required to initiate the precipitation of PbCl2.(Ksp for PbCl2 is 2.40 ×10−4.)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, portedon8644
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 02:30, rileyeddins1010
List four observations that indicate that a chemical reaction may be taking place
Answers: 1

Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1
You know the right answer?
Lead(II) nitrate is added slowly to a solution that is 0.0800 M in Cl− ions. Calculate the concentra...
Questions in other subjects:










Social Studies, 27.08.2019 21:00

![[Pb^{2+}]=3.9 \times 10^{-2}M](/tpl/images/0588/1239/e3715.png)
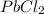 ⇄
⇄ 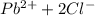
![Ip=[Pb^{2}][2Cl^-]^2=Ksp](/tpl/images/0588/1239/35efe.png)
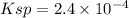
![[Pb^{2+}] = S](/tpl/images/0588/1239/e7380.png)
![[Cl^-]=2S](/tpl/images/0588/1239/41e82.png)
![Ksp=[Pb^{2+}]\times [Cl^-]^2](/tpl/images/0588/1239/02fd6.png)
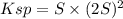
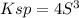
![S=\sqrt[3]{\frac{Ksp}{4} }](/tpl/images/0588/1239/e8aed.png)