
Chemistry, 07.04.2020 21:50 hunteryolanda82
The Kb of weak base B is 1.0 x 10-6. A solution contains 0.75 M B and 0.25 M HB+Cl- (the salt of B with HCl). What is the pH of the solution after 0.05 mol NaOH is added to 1.0 L of the above solution?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 21:30, crystalbyrd79p8imrx
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 23.06.2019 01:30, Dmoney5104
The biomedical technique in which a part of the brain is destroyed with electric current is known as a. electroconvulsive therapy b. prefrontal lobotomy c. bilateral cingulotomy d. tardive dyskinesia
Answers: 2

Chemistry, 23.06.2019 04:10, angelina6836
Which of the following is described by the equation h2o(s)+ heat=h2o(i) a freezing melting condensing evaporating
Answers: 2
You know the right answer?
The Kb of weak base B is 1.0 x 10-6. A solution contains 0.75 M B and 0.25 M HB+Cl- (the salt of B w...
Questions in other subjects:


Social Studies, 24.10.2020 01:00

Mathematics, 24.10.2020 01:00

Biology, 24.10.2020 01:00

Computers and Technology, 24.10.2020 01:00



Mathematics, 24.10.2020 01:00


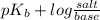
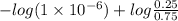
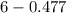
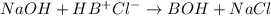
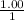 , [Salt] =
, [Salt] = 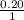
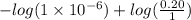 [/tex]
[/tex]

