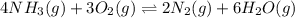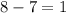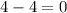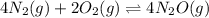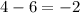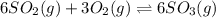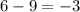Chemistry, 07.04.2020 17:24 19thomasar
In which of the following reactions will Kc= Kp?A) 4 NH3(g) + 3 O2(g) ⇌2 N2(g) + 6 H2O(g)B) 2 SO3(g) + 2 NO(g) ⇌2 SO2(g) + 2 NO2(g)C) 4 N2(g) + 2 O2(g) ⇌4 N2O(g)D) 6 SO2(g) + 3 O2(g) ⇌6 SO3(g)E) None of theabove reactions have Kc= Kp

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:40, alexisbcatlett14
Which statement can best be concluded from the ideal gas law?
Answers: 2

You know the right answer?
In which of the following reactions will Kc= Kp?A) 4 NH3(g) + 3 O2(g) ⇌2 N2(g) + 6 H2O(g)B) 2 SO3(g)...
Questions in other subjects:

History, 26.07.2019 02:00




Mathematics, 26.07.2019 02:00

Health, 26.07.2019 02:00

Computers and Technology, 26.07.2019 02:00

Biology, 26.07.2019 02:00

Mathematics, 26.07.2019 02:00

English, 26.07.2019 02:00

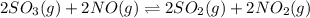
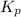 with
with  is given by the formula:
is given by the formula: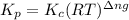
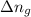 = change in number of moles of gas particles =
= change in number of moles of gas particles = 
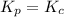 when
when