
Chemistry, 07.04.2020 16:59 shanasia76
A 25.0 g sample of an alloy was heated to 100.0 oC and dropped into a beaker containing 90 grams of water at 25.32 oC. The temperature of the water rose to a final value of 27.18 oC. Neglecting heat losses to the room and the heat capacity of the beaker itself, what is the specific heat of the alloy

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, waterborn7152
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

You know the right answer?
A 25.0 g sample of an alloy was heated to 100.0 oC and dropped into a beaker containing 90 grams of...
Questions in other subjects:


Physics, 19.12.2019 19:31





Health, 19.12.2019 19:31

Mathematics, 19.12.2019 19:31


Engineering, 19.12.2019 19:31

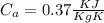
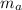 = 25 gm
= 25 gm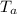 = 100°c = 373 K
= 100°c = 373 K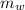 = 90 gm
= 90 gm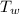 = 25.32 °c = 298.32 K
= 25.32 °c = 298.32 K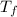 = 27.18 °c = 300.18 K
= 27.18 °c = 300.18 K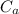 [
[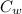 (
(

