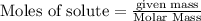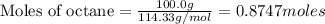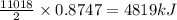Chemistry, 07.04.2020 17:02 cylertroutner
Using the following equation for the combustion of octane, calculate the heat associated with the combustion of 100.0 g of octane assuming complete combustion. The molar mass of octane is 114.33 g/mole. The molar mass of oxygen is 31.9988 g/mole.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:00, princessakosua2
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1


Chemistry, 23.06.2019 00:30, StayPuftMarshadowMan
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1
You know the right answer?
Using the following equation for the combustion of octane, calculate the heat associated with the co...
Questions in other subjects:

Mathematics, 19.02.2020 04:51






World Languages, 19.02.2020 04:52

English, 19.02.2020 04:52


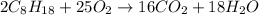 ΔH°rxn =-11018 kJ
ΔH°rxn =-11018 kJ