
Chemistry, 07.04.2020 04:59 tnbankspines
Using your value of Ksp, and starting with an equilibrium system consisting of a saturated solution of calcium hydroxide, predict whether the solubility will increase or decrease it you add a. A. 5.00 mL of 0.100 M HNO3 Ksp = 2.966610-5 b. 5.00 mL of 0.100 M NaOH C. 5.00 mL of 0.100 M Ca(NO3)2

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3


Chemistry, 22.06.2019 18:30, rosie20052019
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 23.06.2019 10:00, kidre96521
State the effect on the concentration of the clo- ion when there is a decrease in the concentration of the oh- ion
Answers: 1
You know the right answer?
Using your value of Ksp, and starting with an equilibrium system consisting of a saturated solution...
Questions in other subjects:


English, 01.07.2019 01:30

Health, 01.07.2019 01:30

Mathematics, 01.07.2019 01:30


Mathematics, 01.07.2019 01:30


Mathematics, 01.07.2019 01:30


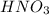 Solubility will increase.
Solubility will increase.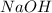 Solubility will decrease.
Solubility will decrease.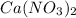 Solubility will decrease.
Solubility will decrease.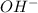 are consumed due to it and the system would respond by reaction shifting towards creation of more
are consumed due to it and the system would respond by reaction shifting towards creation of more 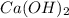 precipitating back. Hence solubility decreases.
precipitating back. Hence solubility decreases.
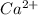 which will be countered by reaction shifting to side that decreases increased
which will be countered by reaction shifting to side that decreases increased 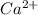 , which happens by some
, which happens by some 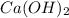 precipitating back. Hence solubility decreases.
precipitating back. Hence solubility decreases.

