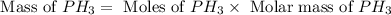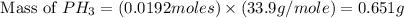Chemistry, 07.04.2020 01:35 harodkdc7910
The reaction of NH3 and O2 forms NO and water. The NO can be used to convert P4 to P4O6, forming N2 in the process. The P4O6 can be treated with water to form H3PO3, which forms PH3 and H3PO4 when heated. Find the mass of PH3 that forms from the reaction of 1.95 g NH3.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, bossboybaker
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 04:00, nikkih1225
Electric charge is what ? a. kinetic energy b. radiation c. discovery d. electricity
Answers: 1


Chemistry, 22.06.2019 15:30, 20cschultz
Which suspect most likely committed the robbery and how do you know
Answers: 2
You know the right answer?
The reaction of NH3 and O2 forms NO and water. The NO can be used to convert P4 to P4O6, forming N2...
Questions in other subjects:


Spanish, 04.07.2019 05:20


SAT, 04.07.2019 05:20

Mathematics, 04.07.2019 05:20




History, 04.07.2019 05:20

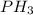 produced from the reaction is, 0.651 grams.
produced from the reaction is, 0.651 grams.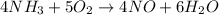
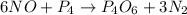
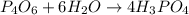
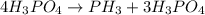
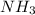
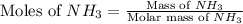
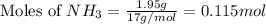
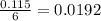 moles of
moles of 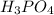
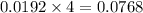 moles of
moles of 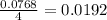 moles of
moles of