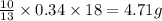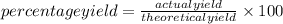Chemistry, 07.04.2020 01:38 vanessa791
Gaseous butane CH3(CH2)2CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. If 1.31g of water is produced from the reaction of 4.65g of butane and 10.8g of oxygen gas, calculate the percent yield of water. Be sure your answer has the correct number of significant digits in it.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, anarosa331hotmailcom
What is driving behind plate tectonics (plate movment)? a) gravity only b) inertia c) convection and gravity d) the sun theres no option for science so i picked chemistry. plz
Answers: 2



Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1
You know the right answer?
Gaseous butane CH3(CH2)2CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2...
Questions in other subjects:

Spanish, 02.02.2021 14:00


Mathematics, 02.02.2021 14:00

English, 02.02.2021 14:00


Physics, 02.02.2021 14:00

Mathematics, 02.02.2021 14:00

Mathematics, 02.02.2021 14:00

Mathematics, 02.02.2021 14:00

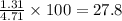
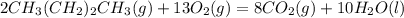
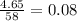
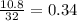
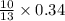 mole of water.
mole of water.