
Chemistry, 06.04.2020 18:02 wilkoASK2919
For the reaction 2A(g) â B(g), the equilibrium constant is Kp = 0.76. A reaction mixture initially contains 4.0 atm of gas (PA = 2.0 atm and PB = 2.0 atm).
Which statement is true of the reaction mixture?
(a) The reaction mixture will proceed toward products.
(b) The reaction mixture is at equilibrium.
(c) The reaction mixture will proceed toward reactants
(d) It is not possible to determine from the information given the future direction of the reaction mixture

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, pandasarecute53
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 09:30, andrejr0330jr
What is the molar mass of potassium nitrate, kno3
Answers: 1

Chemistry, 22.06.2019 11:30, ayoismeisjjjjuan
Which statement best describes the flow of energy in this scenario
Answers: 1
You know the right answer?
For the reaction 2A(g) â B(g), the equilibrium constant is Kp = 0.76. A reaction mixture initially c...
Questions in other subjects:



English, 07.05.2020 00:02



English, 07.05.2020 00:02


Mathematics, 07.05.2020 00:02


Mathematics, 07.05.2020 00:02

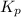
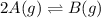
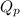 is written as:
is written as: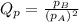

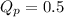
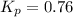
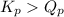 , the reaction will shift towards the right i.e. towards the product side.
, the reaction will shift towards the right i.e. towards the product side.

