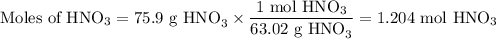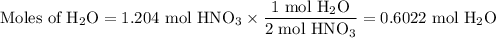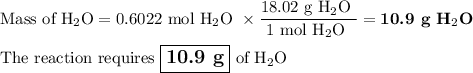Not understanding on how stoichiometry works not sure which side to start on. ex: How many grams of water are required to form 75.9 g of HNO3? Assume that there is excess NO2 present. The molar masses are as follows: H2O = 18.02 g/mol, HNO3 = 63.02 g/mol.
3 NO2(g) + H2O(l) → 2 HNO3(aq) + NO(g)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, 767sebmont
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 22.06.2019 13:50, awesomegamergurl13
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
Not understanding on how stoichiometry works not sure which side to start on. ex: How many grams of...
Questions in other subjects:




Geography, 22.04.2021 22:10

Mathematics, 22.04.2021 22:10





English, 22.04.2021 22:10