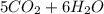Chemistry, 04.04.2020 19:45 gibbss80stu
At 500.K and 1.00 atm pressure, 1.5 liters of pentane gas, C5H12, is mixed with 15 liters of oxygen gas. A complete combustion results. How many liters of WATER vapor, measured at the same temperature and pressure, would be produced after the reaction?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:50, algahimnada
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 22.06.2019 09:00, kcarstensen59070
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1


Chemistry, 22.06.2019 18:30, madmatt873
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1
You know the right answer?
At 500.K and 1.00 atm pressure, 1.5 liters of pentane gas, C5H12, is mixed with 15 liters of oxygen...
Questions in other subjects:



Mathematics, 22.04.2020 22:03

Mathematics, 22.04.2020 22:03




Mathematics, 22.04.2020 22:04


Mathematics, 22.04.2020 22:04

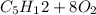 →
→