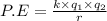Assuming that the distances between the two ions are the same in all cases, which of the following ion pairs has the greatest electrostatic potential energy (i. e., largest in magnitude)? Please explain your answer. a.) Na+ - Cl- b.) Na+ - O-2. c.) Al+3 - O-2. d.) Mg+2-O-2 e.) Na- -Mg+2

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, hannahhoskings6989
What was bohr’s contribution to the planetary model
Answers: 1


Chemistry, 22.06.2019 20:00, jalenevoyles
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 23.06.2019 00:30, mariaramirez110379
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1
You know the right answer?
Assuming that the distances between the two ions are the same in all cases, which of the following i...
Questions in other subjects:


Physics, 29.06.2019 06:30

Mathematics, 29.06.2019 06:30


Arts, 29.06.2019 06:30

Mathematics, 29.06.2019 06:30


English, 29.06.2019 06:30

Geography, 29.06.2019 06:30