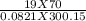A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the tank was found in the morning, the pressure in the tank had dropped to 19.0 atm. If the tank originally held 119.4 moles of oxygen gas, how many moles of gas were left the next morning, assuming the temperature and volume of the rank stayed constant?.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1


Chemistry, 22.06.2019 07:10, angellong94
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 23.06.2019 02:30, elyzarobertson
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the t...
Questions in other subjects:

Social Studies, 23.04.2021 01:30

Spanish, 23.04.2021 01:30





Mathematics, 23.04.2021 01:30

Mathematics, 23.04.2021 01:30


Mathematics, 23.04.2021 01:30