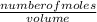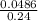Chemistry, 04.04.2020 08:31 Boondiddy9
Suppose that you have 135 mL of a buffer that is 0.360 M in both propanoic acid ( C 2 H 5 COOH ) and its conjugate base ( C 2 H 5 COO − ) . Calculate the maximum volume of 0.240 M HCl that can be added to the buffer before its buffering capacity is lost.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, qwerty8364
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 01:00, deidaralove90
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3

Chemistry, 22.06.2019 17:30, mwest200316
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Suppose that you have 135 mL of a buffer that is 0.360 M in both propanoic acid ( C 2 H 5 COOH ) and...
Questions in other subjects:



Mathematics, 31.10.2020 03:50

Mathematics, 31.10.2020 03:50

English, 31.10.2020 03:50


English, 31.10.2020 03:50

Health, 31.10.2020 03:50