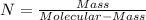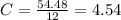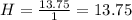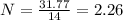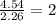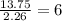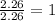Chemistry, 03.04.2020 17:33 andreamartinez7876
(10 points) What is the empirical formula for a compound that is 54.48% carbon, 13.75% hydrogen, and 31.77 % nitrogen?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2


Chemistry, 24.06.2019 01:40, kandiceboggs1
Can someone me with questions 1 and to for masteries plz
Answers: 3

Chemistry, 24.06.2019 05:30, clairajogriggsk
Fill in the blank chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is _ _ ( _ _ ) ._ chemical name chemical formula chemical name chemical formula acetate c2h3o2– nitrite no2– carbonate co32– ammonium nh4+ hypocarbonite co2− cyanide cn– hydrogen carbonate (bicarbonate) hco3– hydroxide oh– chlorite clo2– peroxide o22– hypochlorite clo– phosphate po43– chlorate clo3– hydrogen phosphate hpo42– perchlorate clo4– dihydrogen phosphate h2po4– hydronium h3o+ sulfate so42– permanganate mno4– hydrogen sulfate (bisulfate) hso4– nitrate no3– sulfite so32–
Answers: 1
You know the right answer?
(10 points) What is the empirical formula for a compound that is 54.48% carbon, 13.75% hydrogen, and...
Questions in other subjects:




Mathematics, 06.03.2020 13:54



Medicine, 06.03.2020 13:55

Mathematics, 06.03.2020 13:55


English, 06.03.2020 13:56

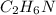 .
.