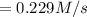Chemistry, 02.04.2020 02:03 recannon02
Consider the reaction: n2(g) 3h2(g) → 2nh3(g) suppose that a particular moment during the reaction, molecular hydrogen is reacting at a rate of 0.0687 m/s. at what rate is molecular nitrogen reacting

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 19:30, Karinaccccc
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 22.06.2019 21:40, fatherbamboo
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
Consider the reaction: n2(g) 3h2(g) → 2nh3(g) suppose that a particular moment during the reaction,...
Questions in other subjects:


Mathematics, 04.10.2019 18:30

Social Studies, 04.10.2019 18:30

Social Studies, 04.10.2019 18:30




History, 04.10.2019 18:30


Biology, 04.10.2019 18:30

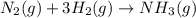
![R=-\frac{1}{1}\frac{d[N_2]}{dt}=-\frac{1}{3}\frac{d[H_2]}{dt}=\frac{1}{2}\frac{d[NH_3]}{dt}](/tpl/images/0576/9841/80d75.png)
![\frac{d[H_2]}{dt}=0.0687 M/s](/tpl/images/0576/9841/7c36a.png)
![\frac{d[N_2]}{dt}=?](/tpl/images/0576/9841/5488c.png)
![-\frac{1}{1}\frac{d[N_2]}{dt}=-\frac{1}{3}\frac{d[H_2]}{dt}](/tpl/images/0576/9841/01276.png)
![\frac{d[N_2]}{dt}=\frac{1}{3}\times 0.0687 M/s](/tpl/images/0576/9841/fd1e5.png)