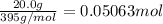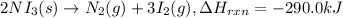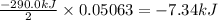Chemistry, 02.04.2020 01:13 wichserc3399
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represented as 2NI3(s)→N2(g)+3I2(g), ΔHrxn=−290.0 kJ Find the change in enthaply when 20.0 g of NI3 decomposes.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, mandy9386
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1


Chemistry, 22.06.2019 13:30, bryce99
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
You know the right answer?
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represe...
Questions in other subjects:





Mathematics, 24.03.2021 18:40

Mathematics, 24.03.2021 18:40


Mathematics, 24.03.2021 18:40

English, 24.03.2021 18:40

Mathematics, 24.03.2021 18:40