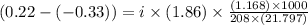Chemistry, 31.03.2020 01:05 raishagibson
Assume that you were assigned BaCl2 in lab. The water in your test tube weighed 21.797 g. Following the procedure in the lab manual, you determined that freezing point of water is 0.02oC. You weighed out 1.168 g of salt and added it to the original test tube, then determined that the freezing point was -0.33oC. Based on these experimental parameters, calculate the van't Hoff factor for BaCl2.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 14:30, hjlhdjfhjh
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 22.06.2019 15:00, raeprince9213
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
Assume that you were assigned BaCl2 in lab. The water in your test tube weighed 21.797 g. Following...
Questions in other subjects:

Business, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

Social Studies, 17.03.2021 23:50

History, 17.03.2021 23:50





History, 17.03.2021 23:50

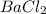 is 1.15
is 1.15
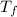 = freezing point of solution =
= freezing point of solution = 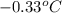
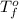 = freezing point of water =
= freezing point of water = 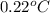
 = freezing point constant of water =
= freezing point constant of water = 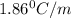
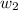 = mass of solute = 1.168 g
= mass of solute = 1.168 g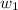 = mass of solvent (water) = 21.797 g
= mass of solvent (water) = 21.797 g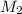 = molar mass of solute = 208 g/mol
= molar mass of solute = 208 g/mol