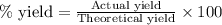Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, Sarahinator04
0.09 moles of sodium sulfate in 12 ml of solution
Answers: 3

Chemistry, 22.06.2019 08:30, MacenParisi
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2


Chemistry, 22.06.2019 14:30, villarrealc1987
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2
You know the right answer?
Assuming an efficiency of 21.20 % , calculate the actual yield of magnesium nitrate formed from 143....
Questions in other subjects:

Biology, 15.04.2020 03:49






Mathematics, 15.04.2020 03:49



 .....(1)
.....(1)

 of magnesium nitrate
of magnesium nitrate