Chemistry, 31.03.2020 00:27 coltonwsmith
Aqueous hydrobromic acid will react with solid sodium hydroxide to produce aqueous sodium bromide and liquid water . Suppose 17. g of hydrobromic acid is mixed with 2.44 g of sodium hydroxide. Calculate the maximum mass of sodium bromide that could be produced by the chemical reaction. Be sure your answer has the correct number of significant digits.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, aleilyg2005
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 10:50, mi364
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

You know the right answer?
Aqueous hydrobromic acid will react with solid sodium hydroxide to produce aqueous sodium bromide an...
Questions in other subjects:



Mathematics, 17.06.2020 22:57

Mathematics, 17.06.2020 22:57


History, 17.06.2020 22:57


Mathematics, 17.06.2020 22:57


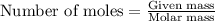 .....(1)
.....(1)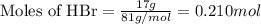
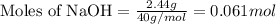

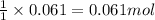 of HBr
of HBr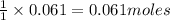 of carbon dioxide
of carbon dioxide