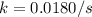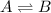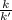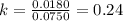Chemistry, 30.03.2020 23:51 eldiamonte
At a given temperature, the elementary reaction A − ⇀ ↽ − B , in the forward direction, is first order in A with a rate constant of 0.0180 s − 1 . The reverse reaction is first order in B and the rate constant is 0.0750 s − 1 . What is the value of the equilibrium constant for the reaction A − ⇀ ↽ − B at this temperature?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1


Chemistry, 23.06.2019 01:00, daniel1480
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
You know the right answer?
At a given temperature, the elementary reaction A − ⇀ ↽ − B , in the forward direction, is first ord...
Questions in other subjects:







Physics, 26.01.2020 20:31



Mathematics, 26.01.2020 20:31