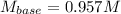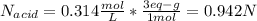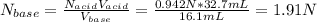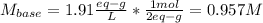Chemistry, 30.03.2020 23:35 ethanyayger
A 16.1 mL sample of Ba(OH)2 is titrated with H3PO4. If 32.7 mL of 0.314 M H3PO4 is needed to reach the endpoint, what is the concentration (M) of the Ba(OH)2 solution? 3 Ba(OH)2(aq) + 2 H3PO4(aq) → Ba3(PO4)2(aq) + 6 H2O(l)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, angelicar1160
How do scientists think that gravity affected the formation of our solar system?
Answers: 1

Chemistry, 22.06.2019 11:40, tatemelliott
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 13:30, kkingstone7062
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1
You know the right answer?
A 16.1 mL sample of Ba(OH)2 is titrated with H3PO4. If 32.7 mL of 0.314 M H3PO4 is needed to reach t...
Questions in other subjects:


Chemistry, 20.08.2021 05:40





Geography, 20.08.2021 05:40


Mathematics, 20.08.2021 05:40

Mathematics, 20.08.2021 05:50