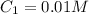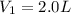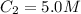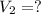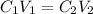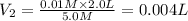The Ksp of barium fluoride, BaF2, is 1.6 x 10-6. A solution of 5.0M NaF is added dropwise to a 2.0L solution that is 0.016 M in Ba2+. When the concentration of fluoride ion exceeds M, BaF2 will precipitate. What volume (in mL) of NaF must be added to cause BaF2 to precipitate? mL

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, timiaparker
What does x represent in the formula for the compound xcl4?
Answers: 2

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 17:00, princessakosua2
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1
You know the right answer?
The Ksp of barium fluoride, BaF2, is 1.6 x 10-6. A solution of 5.0M NaF is added dropwise to a 2.0L...
Questions in other subjects:





Law, 12.11.2020 19:20

Mathematics, 12.11.2020 19:20

English, 12.11.2020 19:20

Mathematics, 12.11.2020 19:20

Mathematics, 12.11.2020 19:20

History, 12.11.2020 19:20

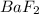 to precipitate.
to precipitate.![[Ba^{2+}]=0.016 M](/tpl/images/0571/5236/69271.png)
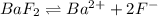
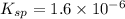
![K_{sp}=[Ba^{2+}][F^-]^2](/tpl/images/0571/5236/2804d.png)
![1.6\times 10^{-6}=[0.016 M]\times [F^-]^2](/tpl/images/0571/5236/5c597.png)
![[F^-]=0.01 M](/tpl/images/0571/5236/09813.png)