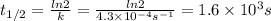Chemistry, 30.03.2020 21:50 karenjjensen
The rate constant, k, for a first-order reaction is equal to 4.3 × 10-4 s-1. What is the half-life for the reaction? The rate constant, k, for a first-order reaction is equal to 4.3 × 10-4 s-1. What is the half-life for the reaction? 1.9 × 103 s 1.2 × 103 s 1.6 × 103 s 3.0 × 10-4 s

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:20, greenbyron88
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 16:30, joshua1255
Find the number of moles of argon in 364g of argon.
Answers: 2

Chemistry, 22.06.2019 21:30, kawaiiblurainbow
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
The rate constant, k, for a first-order reaction is equal to 4.3 × 10-4 s-1. What is the half-life f...
Questions in other subjects:


Mathematics, 15.01.2021 19:20

Mathematics, 15.01.2021 19:20


Spanish, 15.01.2021 19:20


Chemistry, 15.01.2021 19:20



![rate=k \times [A]^{m}](/tpl/images/0571/4200/f39c7.png)