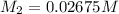Chemistry, 30.03.2020 21:08 studentms5313
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilution. At each stage, 25.00 mL of solution are diluted to 100.00 mL. Determine the concentration of and the number of moles of HNO3 in the final (most dilute, Md5) solution.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, officialgraciela67
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 07:00, coolkid2041
Calculate the number of moles of ethane in 100 grams
Answers: 3

Chemistry, 22.06.2019 07:30, avisconti571
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1
You know the right answer?
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilu...
Questions in other subjects:


Mathematics, 01.09.2019 12:50

Health, 01.09.2019 12:50

History, 01.09.2019 12:50

History, 01.09.2019 12:50


Social Studies, 01.09.2019 12:50

Mathematics, 01.09.2019 12:50


 in the final solution is 0.006688 M and number of moles are 0.00006688
in the final solution is 0.006688 M and number of moles are 0.00006688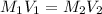
 = molarity of stock solution = 6.847 M
= molarity of stock solution = 6.847 M = volume of stock solution = 25.00 ml
= volume of stock solution = 25.00 ml = molarity of ist dilute solution = ?
= molarity of ist dilute solution = ? = volume of first dilute solution = 100.0 ml
= volume of first dilute solution = 100.0 ml