
Chemistry, 30.03.2020 20:50 thedoragreen
In the laboratory a student combines 26.2 mL of a 0.234 M chromium(III) acetate solution with 10.7 mL of a 0.461 M chromium(III) nitrate solution. What is the final concentration of chromium(III) cation ?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, cxttiemsp021
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 15:20, Tringirl233
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 23.06.2019 00:00, samangelzrose3576
Exit what is the density of an object having a mass of 5.0 g and a volume of 45.0 cm3?
Answers: 1
You know the right answer?
In the laboratory a student combines 26.2 mL of a 0.234 M chromium(III) acetate solution with 10.7 m...
Questions in other subjects:










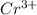 ions in the solution is 0.299 M
ions in the solution is 0.299 M .....(1)
.....(1)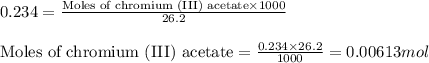
 produces 1 mole of chromium
produces 1 mole of chromium 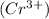 ions and 3 moles of acetate
ions and 3 moles of acetate 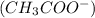 ions
ions

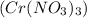 produces 1 mole of chromium
produces 1 mole of chromium 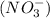 ions
ions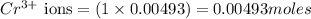
![\text{Molarity of }Cr^{3+}\text{ cations}=\frac{0.01106\times 1000}{36.9}\\\\\text{Molarity of }Cr^{3+}\text{ cations}=0.299M/tex]Hence, the molarity of [tex]Cr^{3+}](/tpl/images/0571/1196/b62db.png) ions in the solution is 0.299 M
ions in the solution is 0.299 M

