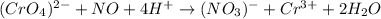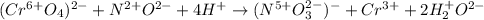Chemistry, 30.03.2020 17:02 naomicervero
CrO42- + NO+ 4H+NO3- + Cr3++ 2H2O In the above redox reaction, use oxidation numbers to identify the element oxidized, the element reduced, the oxidizing agent and the reducing agent. name of the element oxidized: name of the element reduced: formula of the oxidizing agent: formula of the reducing agent:

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3


Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1

Chemistry, 23.06.2019 00:00, vanessacox45
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3
You know the right answer?
CrO42- + NO+ 4H+NO3- + Cr3++ 2H2O In the above redox reaction, use oxidation numbers to identify the...
Questions in other subjects:





Chemistry, 13.07.2019 07:40



Chemistry, 13.07.2019 07:40

Mathematics, 13.07.2019 07:40