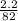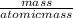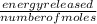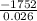A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass of 82.0 g/mol ) is added, it dissolves via the reaction X(s)+H2O(l)→X(aq) and the temperature of the solution increases to 25.5 ∘C . Calculate the enthalpy change, ΔH, for this reaction per mole of X. Assume that the specific heat of the resulting solution is equal to that of water [4.18 J/(g⋅∘C)], that density of water is 1.00 g/mL, and that no heat is lost to the calorimeter itself, nor to the surroundings

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3


Chemistry, 22.06.2019 07:30, deidaraXneji
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1
You know the right answer?
A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass...
Questions in other subjects:




Mathematics, 03.02.2021 17:00






Social Studies, 03.02.2021 17:00