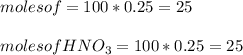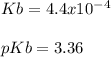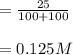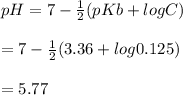A 100 mL sample of 0.25 M CH3NH2(aq) is titrated with a 100 mL of 0.25 M HNO3(aq). Select ALL main components (greater than 0.001 moles, besides H2O) that would be present in the solution after adding HNO3. Kb of CH3NH2 is 4.4 LaTeX: \times×10−4.
OH−
CH3NH2
NO3−
CH3NH3+

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 22:30, arodavoarodavo
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 22.06.2019 22:30, StupidFatChipmunk
What must be in balance for temperatures to remain constant?
Answers: 1

Chemistry, 23.06.2019 10:30, mikemofun9079
Silver is a white metal that is an excellent conductor. silver tarnishes when exposed to air and light. the density of silver is 10.49 g/cm3. the melting point is 962oc and the boiling point is 2000oc. a chemical property of silver is
Answers: 3
You know the right answer?
A 100 mL sample of 0.25 M CH3NH2(aq) is titrated with a 100 mL of 0.25 M HNO3(aq). Select ALL main c...
Questions in other subjects:


History, 21.02.2020 05:22

Mathematics, 21.02.2020 05:22

Mathematics, 21.02.2020 05:22






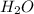 ) that would be present in the solution after adding
) that would be present in the solution after adding 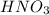 ."
." 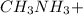 would be present in the solution after adding
would be present in the solution after adding 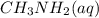 is titrated with a 100 mL of 0.25 M
is titrated with a 100 mL of 0.25 M 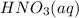 .
.