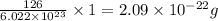a) How many Na^+ ions are present?


Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, rubimachuca1020
Given 7.65 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield?
Answers: 3


You know the right answer?
A sample of sodium sulfite has a mass of 2.80 g.
a) How many Na^+ ions are present?
a) How many Na^+ ions are present?
Questions in other subjects:






Spanish, 04.07.2019 19:00

Business, 04.07.2019 19:00



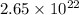
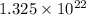
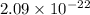 grams
grams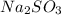 . It is formed by the combination of 2 sodium
. It is formed by the combination of 2 sodium 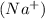 ions and 1 sulfite
ions and 1 sulfite 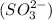 ions
ions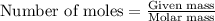

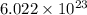 number of particles
number of particles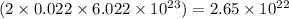 number of sodium ions
number of sodium ions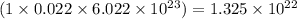 number of sulfite ions
number of sulfite ions