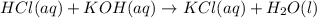During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a flask. The student adds four drops of phenolphthalein to the solution in the flask. The solution is titrated with 0.150 M KOH(aq) until the solution appears faint pink. The volume of KOH(aq) added is 18.5 mL. Complete the equation for the neutralization reaction that occurs during the titration: KOH (aq) + HCl (aq) -->

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 11:40, tatemelliott
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 14:30, lorrainelopez
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
You know the right answer?
During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a fl...
Questions in other subjects:


Social Studies, 18.11.2020 19:20


Business, 18.11.2020 19:20

Health, 18.11.2020 19:20

History, 18.11.2020 19:20



Biology, 18.11.2020 19:20