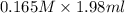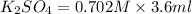Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 15:40, markipler01
What is the study of how matter and energy interact? a. biology b. physics c. planetary science d. chemistry
Answers: 1

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 04:00, heavyhearttim
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2
You know the right answer?
When aqueous solutions of K2SO4 and Pb(NO3)2 are combined, PbSO4 precipitates. Calculate the mass, i...
Questions in other subjects:




Biology, 27.02.2020 22:26

Biology, 27.02.2020 22:26

History, 27.02.2020 22:26



History, 27.02.2020 22:26

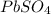 produced is 99.1 g.
produced is 99.1 g.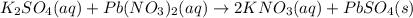
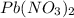 = Molarity × volume
= Molarity × volume