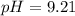Chemistry, 26.03.2020 17:49 seaotter9630
A chemist titrates 90.0 mL of a 0.5870 M acetic acid (HCH3CO2) solution with 0.4794M NaOH solution at 25 °C. Calculate the pH at equivalence. The p Kg of acetic acid is 4.76. Round your answer to 2 decimal places.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, Apple557
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1


Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3
You know the right answer?
A chemist titrates 90.0 mL of a 0.5870 M acetic acid (HCH3CO2) solution with 0.4794M NaOH solution a...
Questions in other subjects:


Medicine, 05.08.2019 02:10




Mathematics, 05.08.2019 02:10


Mathematics, 05.08.2019 02:10

Mathematics, 05.08.2019 02:10

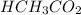 = 0.5870 M
= 0.5870 M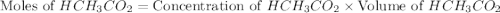
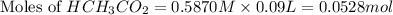
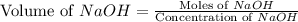
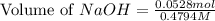
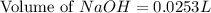
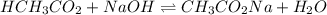
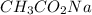 = 0.0528 mol
= 0.0528 mol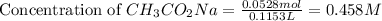
![pH=\frac{1}{2}[pK_w+pK_a+\log C]](/tpl/images/0565/3060/b44e5.png)
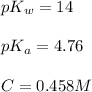
![pH=\frac{1}{2}[14+4.76+\log (0.458)]](/tpl/images/0565/3060/e66f0.png)