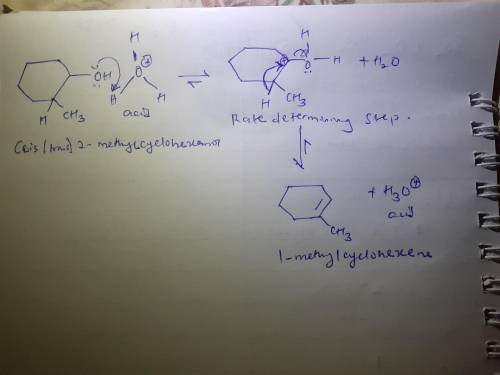
Chemistry, 25.03.2020 17:46 tilly40oooo
The rate-determining step in the acid-catalyzed dehydration of 2-methylcyclohexanol results in the formation of what type of intermediate?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 22.06.2019 21:00, taylorlanehart
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
The rate-determining step in the acid-catalyzed dehydration of 2-methylcyclohexanol results in the f...
Questions in other subjects:





Mathematics, 04.02.2021 18:10

Mathematics, 04.02.2021 18:10

English, 04.02.2021 18:10

History, 04.02.2021 18:10


Mathematics, 04.02.2021 18:10