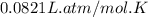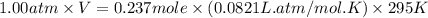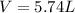Chemistry, 25.03.2020 05:36 lelseymota123
Automobile airbags contain solid sodium azide, NaN 3 , that reacts to produce nitrogen gas when heated, thus inflating the bag. 2 NaN 3 ( s ) ⟶ 2 Na ( s ) + 3 N 2 ( g ) Calculate the value of work, w , for the system if 10.3 g NaN 3 reacts completely at 1.00 atm and 22 ∘ C.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Garciaapril1597
An exothermic reaction is conducted in an insulated calorimeter filled with water. the calorimeter is then sealed so that there is no heat exchanged between the contents of the container and the surrounding air. which of the following statements is true about the reaction?
Answers: 1

Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 12:00, sophiaa23
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
You know the right answer?
Automobile airbags contain solid sodium azide, NaN 3 , that reacts to produce nitrogen gas when heat...
Questions in other subjects:



English, 09.03.2021 21:50






Mathematics, 09.03.2021 21:50

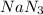
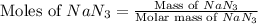
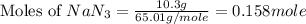
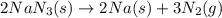
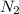
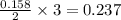 moles of
moles of