Chemistry, 25.03.2020 05:38 codyfore141
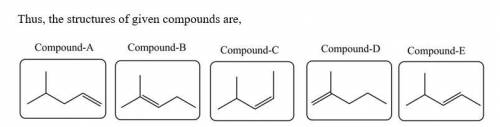
Five isomeric alkenes A–E each undergo catalytic hydrogenation to give 2-methylpentane. The IR spectra of these five alkenes have the following key absorptions (in cm–1): Compound A: 912 (s), 994 (s), 1643 (s), 3077 (m) Compound B: 833 (s), 1667 (w), 3050 (weak shoulder on C–H absorption) Compound C: 714 (s), 1665 (w), 3010 (m) Compound D: 885 (s), 1650 (m), 3086 (m) Compound E: 967 (s), no absorption 1600–1700, 3040 (m) The alkene structures are given below. Identify each compound.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, valeriekbueno
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
You know the right answer?
Five isomeric alkenes A–E each undergo catalytic hydrogenation to give 2-methylpentane. The IR spect...
Questions in other subjects:

English, 01.08.2019 21:00


History, 01.08.2019 21:00


Biology, 01.08.2019 21:00



Health, 01.08.2019 21:00