Chemistry, 25.03.2020 00:32 silvermansachs9189
The reduction of iron(III) oxide to iron during steel-making can be summarized by this sequence of reactions: The net reaction is: Write an equation that gives the overall equilibrium constant in terms of the equilibrium constants and . If you need to include any physical constants, be sure you use their standard symbols, which you'll find in the ALEKS Calculator.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 13:30, citlalli30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2
You know the right answer?
The reduction of iron(III) oxide to iron during steel-making can be summarized by this sequence of r...
Questions in other subjects:



Mathematics, 05.10.2020 14:01

Biology, 05.10.2020 14:01



Social Studies, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01

Health, 05.10.2020 14:01

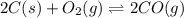 ;
; 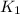
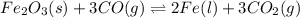 ;
; 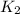
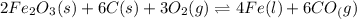 ;
; 
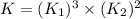
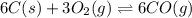 ;
; 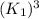
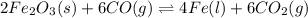 ;
;