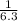Chemistry, 24.03.2020 23:12 ShelleyPSeliger
For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) the equilibrium constant at a certain temperature is 3.10 . At this temperature, calculate the number of moles of NO 2 ( g ) that must be added to 2.30 mol SO 2 ( g ) in order to form 1.00 mol SO 3 ( g ) at equilibrium.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, genyjoannerubiera
Si una estrella no tiene paralaje medible, ¿qué puedes inferir?
Answers: 1

Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 23.06.2019 04:20, milkshakegrande101
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 05:00, jjoyner
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2
You know the right answer?
For the chemical equation SO 2 ( g ) + NO 2 ( g ) − ⇀ ↽ − SO 3 ( g ) + NO ( g ) the equilibrium cons...
Questions in other subjects:

English, 09.03.2021 17:20




Chemistry, 09.03.2021 17:20


Social Studies, 09.03.2021 17:20

Mathematics, 09.03.2021 17:20

Mathematics, 09.03.2021 17:20

Mathematics, 09.03.2021 17:20

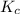 =
= ![\frac{[SO_{3} ][NO]}{[SO_{2}][NO_{2} ]}](/tpl/images/0561/8606/bc0fb.png)
![\frac{(1.00)(1.00)}{(2.30) [NO_{2} ]}](/tpl/images/0561/8606/3f85b.png) At equilibrium [SO₃] = [NO]
At equilibrium [SO₃] = [NO]