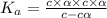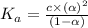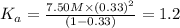Chemistry, 24.03.2020 22:32 allisonlillian
Nitric acid (HNO3) is a strong acid that is completely ionized in aqueous solutions of concentrations ranging from 1% to 10% (1.50 M ). However, in more concentrated solutions, part of the nitric acid is present as un-ionized molecules of HNO3. For example, in a 50% solution (7.50 M ) at 25°C, only 33% of the molecules of HNO3 dissociate into H+ and NO3–. What is the value of Ka for HNO3?

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1
You know the right answer?
Nitric acid (HNO3) is a strong acid that is completely ionized in aqueous solutions of concentration...
Questions in other subjects:

Mathematics, 19.07.2019 08:30


History, 19.07.2019 08:30

Health, 19.07.2019 08:30




History, 19.07.2019 08:30

Biology, 19.07.2019 08:30

SAT, 19.07.2019 08:30

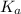 of the nitric acid is 1.2.
of the nitric acid is 1.2.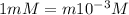
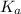
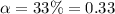
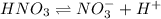
![K_a=\frac{[NO_3^{-}][H^+]}{[HNO_3]}](/tpl/images/0561/7216/4e976.png)