
Chemistry, 24.03.2020 18:13 belladaviau1338
Iron(II) can be oxidized by an acidic K2Cr2O7 solution according to the net ionic equation: Cr2O72− + 6Fe2+ + 14H+ → 2Cr3+ + 6Fe3+ + 7H2O If it takes 35.5 mL of 0.0250 M K2Cr2O7 to titrate 25.0 mL of a solution containing Fe2+, what is the molar concentration of Fe2+ in the original solution?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, hannah5143
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3


You know the right answer?
Iron(II) can be oxidized by an acidic K2Cr2O7 solution according to the net ionic equation: Cr2O72−...
Questions in other subjects:

Mathematics, 14.10.2020 18:01



Mathematics, 14.10.2020 18:01

Spanish, 14.10.2020 18:01



History, 14.10.2020 18:01


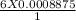 = 0.005325 mole of Fe²
= 0.005325 mole of Fe²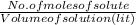
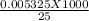 = 1.33 molar
= 1.33 molar

