
Chemistry, 24.03.2020 17:41 adriantheclashroyaln
A 32.5 g iron rod, initially at 22.4 ∘C, is submerged into an unknown mass of water at 63.0 ∘C, in an insulated container. The final temperature of the mixture upon reaching thermal equilibrium is 59.7 ∘C. Part A What is the mass of the water? Express your answer to two significant figures and include the appropriate units.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:00, MrSavannahCat
Which produce would best increase the amount of heat energy that is actually gained by calorimeter b
Answers: 1


Chemistry, 22.06.2019 12:30, johnsont8377
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1
You know the right answer?
A 32.5 g iron rod, initially at 22.4 ∘C, is submerged into an unknown mass of water at 63.0 ∘C, in a...
Questions in other subjects:





History, 19.10.2019 05:10





 = 39.18 gm
= 39.18 gm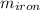 = 32.5 gm
= 32.5 gm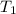 = 22.4°c = 295.4 K
= 22.4°c = 295.4 K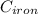 = 0.448
= 0.448 
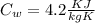
 = 336 K
= 336 K  = 59.7°c = 332.7 K
= 59.7°c = 332.7 K (
(


