
Chemistry, 24.03.2020 16:38 catelinboog04
Ammonium perchlorate is a powerful solid rocket fuel, used in the Space Shuttle boosters. It decomposes into nitrogen gas, chlorine gas, oxygen gas and water vapor, releasing a great deal of energy. Calculate the moles of chlorine produced by the reaction of of ammonium perchlorate. Be sure your answer has a unit symbol, if necessary, and round it to significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, kristieroth1
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 04:30, ajsoccer1705
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3


Chemistry, 22.06.2019 11:40, arlabbe0606
Effect of rotenone and antimycin a on electron transfer rotenone, a toxic natural product from plants, strongly inhibits nadh dehydrogenase of insect and fish mitochondria. antimycin a, a toxic antibiotic, strongly inhibits the oxidation of ubiquinol. (a) explain why rotenone ingestion is lethal to some insect and fish species. (b) explain why antimycin a is a poison. (c) given that rotenone and antimycin a are equally effective in blocking their respective sites in the electron-transfer chain, which would be a more potent poison? explain.
Answers: 3
You know the right answer?
Ammonium perchlorate is a powerful solid rocket fuel, used in the Space Shuttle boosters. It decompo...
Questions in other subjects:

Geography, 18.01.2020 23:31




Mathematics, 18.01.2020 23:31

Physics, 18.01.2020 23:31

Mathematics, 18.01.2020 23:31

Mathematics, 18.01.2020 23:31

Biology, 18.01.2020 23:31

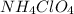 is a powerful solid rocket fuel, used in the Space Shuttle boosters. It decomposes into nitrogen gas, chlorine gas, oxygen gas and water vapor, releasing a great deal of energy. Calculate the moles of chlorine produced by the reaction of 2.5 mol of ammonium perchlorate. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.
is a powerful solid rocket fuel, used in the Space Shuttle boosters. It decomposes into nitrogen gas, chlorine gas, oxygen gas and water vapor, releasing a great deal of energy. Calculate the moles of chlorine produced by the reaction of 2.5 mol of ammonium perchlorate. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.
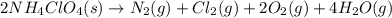
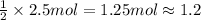 of chlorine gas
of chlorine gas

